Humanness of Protein Therapeutics in the Age of De Novo Protein Design
Does “humanness” make therapeutics safer? Not really.
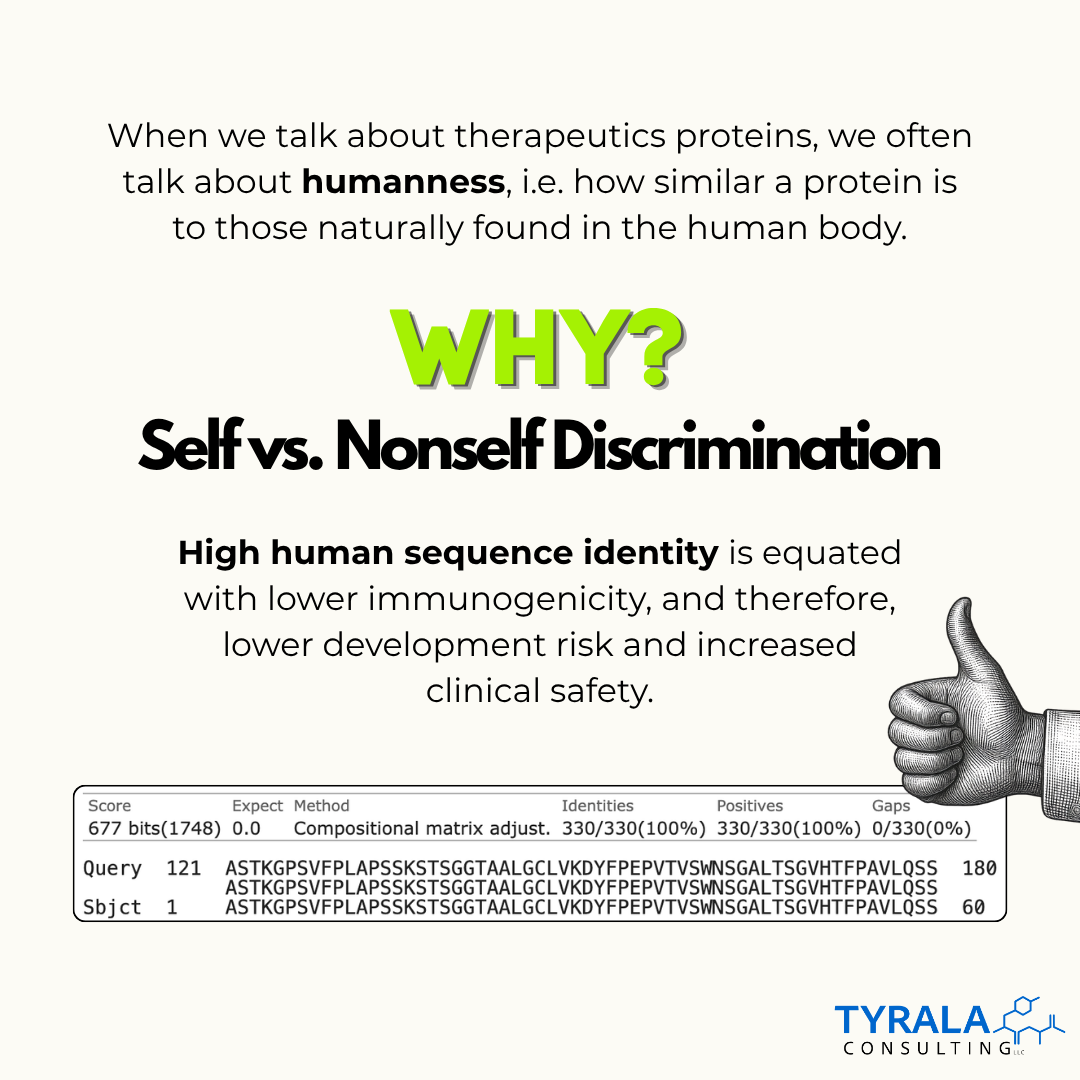
High sequence identity to human proteins has long been viewed as a measure of therapeutic safety with the assumption that high humanness would reduce immunogenicity risk and lower the chances of complications in the clinic.
Humanness isn't the whole picture, though, and it's actually a pretty limiting approach.
Immune responses still occur against highly "human” therapeutics because the immune system and its subsequent response isn't based solely on humanness. So when we design for safety and efficacy, we should focus on how the immune system actually works and what criteria need to be met for an immune response to occur:
→ Uptake and processing of the therapeutic into peptides by APCs
→ MHCII presentation of these peptides
→ Recognition of the MHCII-peptide complex and activation of CD4+ T-cells
→ Sequence homology to common bacterial and viral proteins
Move beyond “humanness.”
Focus on drivers of immune recognition.
This shift in approach is foundational for the design of the next generation of protein therapeutics!